| Video Discription |
Chlorofluorocarbons
Disclaimer: Additional Information That Is Not In Video
Chlorofluorocarbons (CFCs) and Ozone Depletion
The depletion of the ozone layer, caused primarily by chlorofluorocarbons (CFCs), is one of the most pressing environmental issues of our time. In this article, we'll explore what CFCs are, how they contribute to ozone depletion, and the effects of this depletion on the environment and climate.
What are Chlorofluorocarbons (CFCs)?
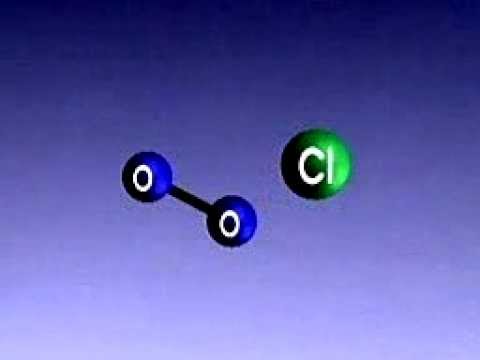
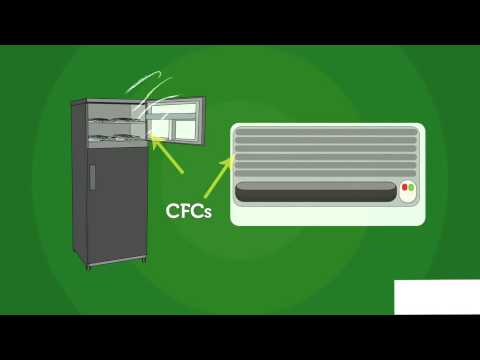
Chlorofluorocarbons, or CFCs, are man-made compounds composed of carbon, chlorine, and fluorine atoms. They were first developed in the early 20th century for use as refrigerants, propellants in aerosol sprays, solvents, and foam-blowing agents due to their stability, low toxicity, and non-flammability.
How do CFCs Deplete the Ozone Layer?
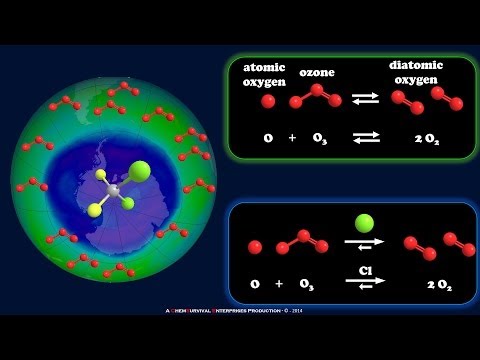
When CFCs are released into the atmosphere, they eventually drift upward into the stratosphere, where they are broken down by ultraviolet (UV) radiation from the sun. This process releases chlorine atoms, which then react with ozone (O3) molecules, leading to their destruction. A single chlorine atom can destroy thousands of ozone molecules in a catalytic chain reaction, resulting in the formation of an ozone hole.
The Effects of Ozone Depletion
Increased UV Radiation: Ozone depletion allows more UV radiation to reach the Earth's surface, posing serious health risks to humans, animals, and plants. UV radiation is linked to cataracts, immune system suppression, and damage to marine ecosystems.
Climate Change: Ozone depletion can also contribute to climate change by altering atmospheric circulation patterns and affecting the distribution of heat and moisture around the globe. This can lead to changes in weather patterns, sea levels, and agricultural productivity.
Ecosystem Disruption: UV radiation can directly harm phytoplankton, which form the base of the marine food chain, leading to cascading effects throughout aquatic ecosystems. Ozone depletion can also affect terrestrial ecosystems by reducing crop yields, disrupting plant growth, and altering species distributions.
The History of Ozone Depletion
The discovery of the ozone hole over Antarctica in the 1980s raised international awareness of the threat posed by CFCs to the ozone layer. Scientists Sherwood Rowland and Mario Molina were awarded the Nobel Prize in Chemistry in 1995 for their research on the role of CFCs in ozone depletion. Their work led to the adoption of the Montreal Protocol in 1987, an international treaty aimed at phasing out the production and use of ozone-depleting substances, including CFCs.
What Can We Do to Address Ozone Depletion?
Phase Out Ozone-Depleting Substances: Continued efforts to phase out the production and use of CFCs and other ozone-depleting substances are crucial for protecting the ozone layer and mitigating its effects on the environment and climate.
Promote Alternatives: Encourage the development and use of ozone-friendly alternatives to CFCs in refrigeration, air conditioning, aerosol products, and foam insulation. These alternatives, such as hydrofluorocarbons (HFCs) and hydrochlorofluorocarbons (HCFCs), have lower ozone-depletion potentials and shorter atmospheric lifetimes.
Raise Awareness: Educate the public about the importance of protecting the ozone layer and the role that individuals can play in reducing their contribution to ozone depletion. Simple actions, such as recycling, reducing energy consumption, and using eco-friendly products, can help minimize the release of ozone-depleting substances into the atmosphere.
Conclusion: Protecting the Ozone Layer for Future Generations
The depletion of the ozone layer due to chlorofluorocarbons and other ozone-depleting substances is a global environmental challenge that requires urgent action. By understanding the causes and effects of ozone depletion and taking meaningful steps to address it, we can protect the ozone layer and safeguard the health of our planet for future generations. Let us all work together to ensure a healthy and sustainable environment for all.
[3Cwbm3W9DF8] |